 /GettyImages-4751580871-b0a7b670482e49bd8197aa5e513ffc83.jpg)
“Resonance Effect or Mesomeric Effect – Definition & Types of Resonance effect.” JEE Class 11-12, Byjus Classes, 17 Feb. “Mesomeric effect.” Wikipedia, Wikimedia Foundation, 16 Sept. The main difference between resonance and mesomeric effect is that resonance occurs due to the interaction between lone electron pairs and bond electron pairs whereas mesomeric effect occurs due to the presence of substituent groups or functional groups. Resonance and mesomeric effect are two concepts that are used to describe the stabilization of molecules via delocalization of electrons throughout the molecule. Mesomeric Effect:Mesomeric effect can be found as positive mesomeric effect and negative mesomeric effect. 
Resonance: Resonance can be found as positive resonance effect and negative resonance effect. Mesomeric Effect:Mesomeric effect occurs due to the presence of substituents/ functional groups or conjugated systems. Resonance: Resonance occurs due to the presence of lone pairs adjacent to double bonds. Mesomeric Effect: Mesomeric effect is the stabilization of a molecule with the use of different functional groups or substituents. Resonance: Resonance is the concept that describes the interaction between lone electron pairs and bond electron pairs of a molecule that eventually determines the chemical structure of that molecule. Difference Between Resonance and Mesomeric Effect Definition This occurs for the stabilization of the molecule. It is the delocalization of the pi bond electron pairs. 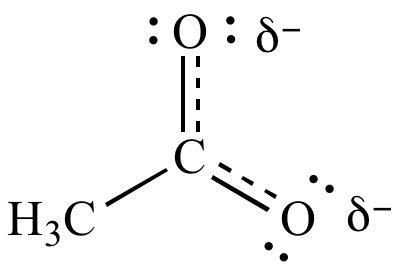
In conjugated systems (molecules having alternating double bonds), the mesomeric effect can be moved along the system. But the actual structure of the molecule is a hybrid of all possible resonance structures.įigure 2: Stabilization of Nitrobenzene through Positive Mesomerism A molecule can have several resonance forms depending on the number of lone electron pairs and pi bonds. The interaction between lone electron pairs on atoms and the pi electron bond pairs of adjacent chemical bonds results in resonances. The resonance of molecules causes the polarity of molecules. This effect can be observed in molecules having double bonds. Resonance is the concept that describes the interaction between lone electron pairs and bond electron pairs of a molecule that eventually determine the chemical structure of that molecule. Key Terms: Bond Electron Pair, Functional Group, Lone Electron Pair, Mesomeric Effect, Negative Mesomeric Effect, Negative Resonance Effect, Polarity, Positive Mesomeric Effect, Positive Resonance Effect, Resonance Effect What is the Difference Between Resonance and Mesomeric Effect Mesomeric effect is the effect of substituents or functional groups on chemical compounds. Resonance is the effect that describes the polarity of a molecule that is induced by interaction between lone electron pairs and bond electron pairs. Resonance and mesomeric effects in molecules determine the exact chemical structure of the molecule. Main Difference – Resonance vs Mesomeric Effect 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
